Managing rare diseases: examples of national approaches in Europe, North America and East Asia
Abstract
Around 4% of the global population suffers from a rare disease. Apart from the medical aspect, economic, organisational, and political approaches remain key aspects when it concerns the evolution of the world of rare diseases. We review here the principal specific national initiatives and organisations in Europe, North America and East Asia. Thereafter, we propose the outlines of a possible optimal approach, inspired by the successes of the individual national organisations. This work should be taken into account in the definition of large scale multi-national rare diseases programs, such as the European Joint Programme on Rare Diseases.
Keywords
INTRODUCTION
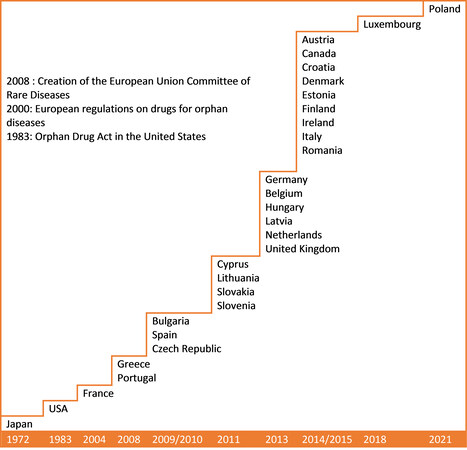
Three hundred million people worldwide suffer from rare diseases, which is equivalent to 4% of the global population. Thirty million patients in Europe are concerned. The first countries to implement a healthcare policy on orphan diseases were Japan with the Nan-Byo (Nan-Byo means “difficult + illness” in Japanese) Consortium in 1972, and the United States with the Orphan Drug Act in 1983. France followed in 2004 with its National Plan for Rare Diseases, setting an example for other European countries and Canada [Figure 1]. Today, among European Union Member States, only Malta and Sweden have no plan in place for rare diseases.
Figure 1. Implementation of national plans and strategies dedicated to rare diseases and their management in Europe, North America and East Asia.
This study describes the national State-coordinated transdisciplinary approaches in place to support patients with rare diseases and oversee their access to healthcare in Europe, North America and East Asia. It then seeks to identify efficient initiatives that could be replicated in order to bring together all the stakeholders in the rare disease health system. An approach of this type would benefit patients and accelerate the development of breakthrough innovations. Given that the purpose of this paper is to study the initiatives and organisations specific to each country, it will not look closely at the organisation of this sector at the European Commission level, nor at the association level. However, considering their essential role, these two levels have to be mentioned:
Since 2008, the European Commission (EC) (The European Commission is the European Union’s politically independent executive arm. It is responsible for drawing up proposals for new European legislation, and it implements the decisions of the European Parliament and the Council of the European Union) has been active in the field of rare diseases, making it a priority of the Commission, even if Europe was already involved in rare diseases, especially with the implementation of the European regulation (No 141/2000) on orphan medicinal products in 2000. The commitment of the EC notably led to the creation in 2009 of the European Committee of Experts on Rare Diseases, which aimed to help the EC with the preparation and implementation of Community activities in the field of rare diseases. This commitment also allowed the funding of shared tools for European Member States, such as the European Reference Networks (ERNs), networks of centres of expertise, healthcare providers and laboratories organised across borders; the European Joint Programme on Rare Diseases (EJPRD), a programme aiming to create an effective rare diseases research ecosystem for progress, innovation and for the benefit of everyone with a rare disease; the European Platform on Rare Diseases Registration (EU RD Platform), platform making data from rare disease registries searchable at a European Union (EU) level and standardising data collection and exchange in order to solve the problem of fragmentation of rare disease data; and the European Rare Disease Research Coordination and Support Action Consortium (ERICA) in 2021, platform composed of a consortium of the 24 ERNs, integrating their research and innovation capacity. A whole paper could be written on the commitment of the European Commission, its work in bringing players together, and the diversity of the programmes funded in the field of rare diseases.
Patient organisations are defined as non-profit organisations which are patient-focused, and whereby patients and/or carers (the latter when patients are unable to represent themselves) represent a majority of members in governing bodies. These organisations provide moral, practical, financial, social and legal support to patients and their families, but they are also able to contribute to research funding and are important partners for health professionals, decision-makers and pharmaceutical laboratories.
METHOD
The selection of the countries included was firstly made on a geographical criterion. Indeed, we limited the work to Europe, East Asia, and North America.
In Europe, we decided to present an exhaustive overview of the national approaches present in the different countries. They were then selected according to the availability and accessibility of the information concerning the national approaches to rare diseases.
In North America and East Asia, the countries were selected using arbitrary criteria. Indeed, Japan and the United States were selected because they represent the first countries to implement a national approach for rare diseases, while Canada was selected because it is considered a pioneer in gene identification.
All the data used hereafter are obtained from the legal texts and from the organisations involved in the different national approaches.
RESULTS
Approaches to rare diseases
European countries
All the European countries mentioned below use the same definition for rare diseases. This definition is provided by the European Medicines Agency (EMA): “a disease is considered rare if fewer than five in 10,000 people have it”.
1. Germany: Since 2020, Germany has stepped up its commitment to research in the field of rare diseases.
(a). The German National Action League for People with Rare Diseases (NAMSE) was set up by the Federal Ministry of Health (BMG) in 2010. It is jointly coordinated by the BMG, the Federal Ministry of Education and Research (BMBF) and the National Alliance for Chronic Rare Diseases (ACHSE). NAMSE is responsible for the implementation, coordination and follow-up of the rare disease plan. ACHSE brings together 28 partners, including the Federal Ministry of Labour and Social Affairs, the German Medical Association and the National Health Insurance Association[1].
As part of this organisation, the German plan for rare diseases came into force in August 2013.
(b). Thirty-six centres of expertise collaborate with a network of 11 research centres, led by a Board of Directors.
(c). The se-Atlas platform for healthcare providers and patients. This platform stores data that will help to improve diagnosis as well as facilitate innovations[2]. In February 2020, the BMBF launched CORD-MI, a two-year project with a funding of €6 million. Led by the Institute of Health in Berlin, the purpose of the project is to collect and structure data relating to rare diseases as part of a digital network. This will then be incorporated into the BMBF Medical Informatics Initiative (MII). The MII was set up to develop an infrastructure for the integration of all patient clinical data, regardless of the field. By late 2022, the BMBF will have invested around €180 million in the MII[3].
(d). Alongside these commitments, the BMBF has announced plans from 2021 to invest €3 million every year in a new centre for neurodegenerative diseases (DZNE) in Ulm. This takes the number of DZNEs to ten (Source: French Embassy in Germany, Scientific Department, 4/5/2018.).
(e). Further, in January 2021, the BMBF announced the launch of an independent Fraunhofer research centre dedicated to Translational Medicine and Pharmacology (Fraunhofer ITMP)[4,5]. With three research sites in Frankfurt, Hamburg and Göttingen, the Fraunhofer ITMP will develop innovative solutions in collaboration with industry and with the support of government funding. Its objectives are to transfer innovations to medical applications and to train future leaders in the field of biomedical research. Pre-clinical and clinical research will be carried out on the basis of interdisciplinary collaboration) (The Fraunhofer Society is a German organisation dedicated to applied research. Funded by the government and private partners, it has 74 institutes across Germany. It is second only to the United States in its field of activity.)
Between 2003 and 2018, the BMBF invested €107 million in the field of rare diseases.
2. Spain: SpainUDP is Spain’s main programme for undiagnosed rare diseases.
In Spain, the rare diseases programme is led by the Carlos III Health Institute (ISCIII).
The ISCIII is a public health research institute founded in 1986 to promote research into biomedicine and health sciences and to develop and steer the scientific and technical strategies of the Spanish national health system. Located on two campuses in Madrid, it helps to promote research and innovation in the health sciences sector. It also provides scientific and technical services and educational programmes[6].
Within the ISCIII, the Institute for Research into Rare Diseases (IIER) is studying the mechanisms underpinning the origin and progression of rare diseases and congenital malformations. The goal of the research projects is to develop innovative treatments.
The IIER is developing several translational programmes:
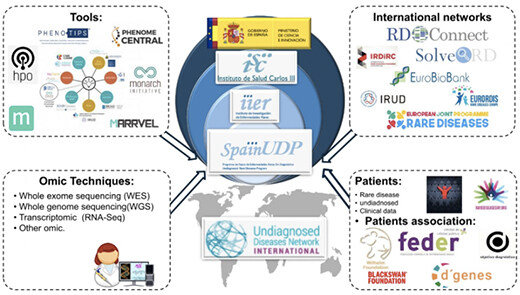
(a). The Undiagnosed Rare Diseases Programme - SpainUDP - was set up to address the high number of consultations for undiagnosed rare diseases. SpainUDP offers patients a multidisciplinary approach. In 2014, the SpainUDP programme became part of the Undiagnosed Diseases Network International (UDNI), a global consortium of research centres and institutes. The partners and tools of SpainUDP are shown in Figure 2.
(b). The Rare Disease Patient Registry (SpainRDR) provides healthcare professionals, researchers, patient groups and their families with extensive information on the number and geographical distribution of people affected by rare diseases in Spain.
SpainRDR is co-funded by the ISCIIII, the Regional Health Departments and the Ministry of Health and Social Services. Its objective is to increase the visibility of rare diseases and support decision-making in order to ensure adequate health planning and resource allocation.
SpainRDR works with hospitals, a network of scientists, patient organisations, and the pharmaceutical industry.
(c). The National Biobank for Rare Diseases (BioNER) provides the scientific community with a catalogue of biological samples associated with clinical and epidemiological information on these diseases.
(d). The Human Genetic Area (AGH) includes a Genetic Diagnosis Service offering services for the diagnosis of the various genetic diseases identified. This centre is equipped with state-of-the-art technologies such as high-throughput sequencing and large-scale data analysis.
3. France: a pioneer in Europe with the first Rare Disease Plan in 2004. Since this date, France has maintained its leadership.
Rare diseases are a priority for France.
The Public Health Act of 2004 led to the creation of a national plan for rare diseases (PNMR), which has been renewed twice (PNMR2 and PNMR3). As part of PNMR3, the State oversees healthcare and research activities. For these purposes, it is represented by the Ministry of Solidarity and Health and the Ministry of Higher Education, Research and Innovation.
These initiatives have made it possible to accelerate patient care, boost teaching and training and drive progress in research and innovation. They are organised as follows:
(a). Nearly 400 Reference Centres for Rare Diseases (CRMRs) with a coordinating site and one or more constituent sites
A CRMR brings together a highly specialised hospital-based team with proven expertise in a particular rare disease or group of rare diseases. Each CRMR develops activities in the fields of prevention, healthcare, teaching and research. The medical team includes paramedical, psychological, medico-social, educational and social skills experts. It also forms partnerships with patient organisations. A CRMR is a multi-site entity. It is an expert referral centre of a regional, interregional, national and even international scope.
(b). 23 National Rare Disease Health Networks (FSMRs)
The FSMRs were set up in 2014-2015 as part of PNMR2. They each cover a broad but coherent field of rare diseases. Each FSMR groups the stakeholders involved in a given area, including caregivers, researchers, patient representatives and manufacturers. Each FSMR pursues three goals: better healthcare, research and education, training and information.
(c). At the same time, the French National Institute for Health and Medical Research (INSERM) is coordinating the European Joint Programme on Rare Diseases (EJPRD), a flagship programme co-funded by the EU, with the purpose of structuring the Rare Disease ecosystem in European and international levels.
(d). The BaMaRa national register, developed by the BNDMR (National Data Bank for Rare Diseases), has now reached the deployment stage. This process involves staff training, data entry checks, data exhaustiveness checks, administrative follow-up and harmonisation of coding.
Connected to the National Health Data System (SNDS), the BaMaRa register aims to develop medical-economic knowledge on rare diseases and to conduct epidemiological studies in order to better assess the prevalence and incidence of rare diseases.
(e). Orphanet
Created in France in 1997, Orphanet was initially developed to gather scarce information concerning rare diseases to improve the diagnosis and the treatment of the patients. Orphanet has now become a network of 41 countries throughout Europe and the world and represents the reference source of information on rare diseases. The objectives of Orphanet are to provide a common language in rare diseases with the Codes ORPHA to have a better comprehension between the actors; provide high-quality data and expertise on rare diseases to allow equal access to knowledge, and guide users in the field of rare diseases; contribute to a better understanding of rare diseases.
(f). The French national programme on Rare Disease Cohorts, RaDiCo, aims to establish national and international cohorts of patients with rare diseases. The objective is to better describe the diseases, establish phenotype/genotype correlations, understand the physiopathology, identify new therapeutic targets and evaluate the medico-economic and societal impact of the diseases.
PNMR3 (2018-2022) has a budget of €778 million, most of which (€597 million) is dedicated to the CRMRs. The initiatives in place include collaborative work with the France Genomic Medicine 2025 plan to facilitate access to very-high-throughput sequencing platforms, in accordance with a scale based on the clinical situation.
Finally, France has internationally renowned research institutes involving public and private partners:
- the Imagine Foundation was set up to conduct a project of medical and scientific excellence by organising, structuring and developing research, healthcare and teaching activities in the field of genetic diseases affecting both children and adults;
- I-Motion is a clinical trial centre dedicated to children with neuromuscular diseases;
- The Institute of Myology promotes the existence, recognition and growth of myology as a discipline in its own right. The muscular system is a true model of innovation for scientific and medical research;
- The NeuroMyoGene Institute is dedicated to studying the physiopathology of the muscular and nervous system, with an opening in clinical and translational research;
- The French Foundation for rare diseases is dedicated to the coordination and acceleration of research on all rare diseases.
4. United Kingdom (UK): an ambitious programme of government investment launched in 2012 has created a highly structured and efficient system that also includes private-sector partners.
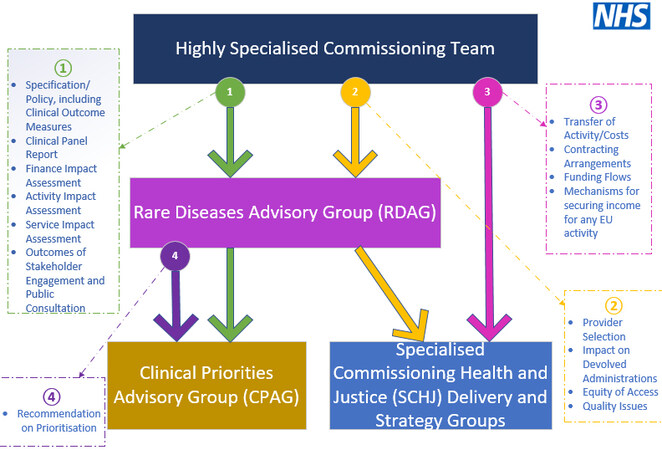
(a). Within the National Health Service (NHS), the Highly Specialised Services (HSSs) are a network of 76 centres specialising in rare diseases[7]. The HSSs place emphasis on patient care and empowerment, with each centre treating no more than 500 patients per year. For the 2018/2019 financial year, the NHS provided €19.8 billion for the running of the 76 centres (2019 UK NHS HSS Report). The governance of the Rare Diseases Advisory Group is described in Figure 3.
Figure 3. Governance of the Rare Diseases Advisory Group (Terms of reference, Rare Diseases Advisory Group, March 2020).
(b). At the same time, 13 genomic medicine centres were set up in 2019 with the participation of 85 hospitals. Their aim is to sequence 50,000 genomes by 2023.
These initiatives are coordinated with the Cell and Gene Therapy Catapult launched in April 2012.
(c). The catapult centre[8]
The nine Catapult Centres are private, independent organisations. Like Germany’s Fraunhofer research centres, they have a mixed-funding model based on an equal split between:
- Grants awarded to each centre by Innovate UK (State funding and innovation body), primarily for purposes of infrastructure and the development of expertise. This grant can total up to €12 million per centre;
- R&D contracts and services, financed by the private sector;
- Grants obtained through calls for proposals from funding agencies.
The Cell and Gene Therapy Catapult (CGT Catapult)[9] was set up to drive progress in gene and cell therapy at UK and international levels. Its mission is to:
- Conduct clinical trials;
- Deliver an infrastructure equipped to the standards required by Good Manufacturing Practices (GMPs);
- Provide expertise on regulatory issues (shorter start-up times for clinical trials);
- Set up national and international collaborative projects;
- Gain access to grants and funding.
Located in London, the CGT Catapult has a process development laboratory with dedicated facilities for clinical trials and reproducibility studies. Further, the CGT Catapult site in Stevenage, which opened in 2018 for a cost of €92 million (of which €4 million was funded by the European Union), is an industrial-scale manufacturing centre. This centre is dedicated to manufacturing gene and cell therapy products in collaboration with industry[10].
The CGT Catapult has 64 public and private partners, including UK universities and manufacturers (40 in all), as well as a number of British and international organisations. The main achievements of the CGT Catapult since 2013 are as follows:
- Nearly €3 billion in industrial investment;
- 26 production units meeting GMP standards;
- 47 companies on campus;
- Over 90 therapies at the development stage;
- 60% of therapies developed in the UK in this field are based on a collaboration with the CGT Catapult;
- A 45% increase in clinical trials.
- In 2019/2020, the GCT Catapult allowed to achievement of:
- 108 projects;
- €7.8 million investment in gene and cell therapy;
- 28 phase III clinical trials;
- 10 spin-outs;
- 7 drugs reimbursed for patients;
- €349 million invested by industry in rare diseases;
- Over 3000 jobs.
North America
5. Canada: globally renowned expertise in gene identification
In Canada, the definition considered a rare disease is a disease that affects fewer than 5 in 10,000 Canadians.
Although Canada only developed a National Plan for Rare Diseases in 2015, it has developed internationally recognised expertise in identifying the genes involved in rare diseases.
(a). Set up in 2000, Genome Canada is a not-for-profit organisation supported by the Canadian Institute of Health Research, Institute of Genetics (CIHR IG)[11]. It coordinates federal and provincial studies through six genome centres with the following missions:
- Develop ties between public and private players;
- Invest in major scientific and technological projects.
In 2017, for example, Genome Canada invested €7 million in the Silent Genome Project led by researchers from the University of British Columbia, whose purpose is to create a database of genetic variations in indigenous populations around the world (Northern health indigenous health 28/2/2018)[12].
(b). The Rare Diseases Models and Mechanisms (RDMMs) is a national network coordinating research projects to gain a clearer understanding of the biology underlying rare diseases in children. It is funded by Genome Canada[13]. Present across Canada, the RDMMs are managed by the Maternal Infant Child & Youth Research Network (MICYRN), which brings together 23 networks of experts and 20 hospitals and academic research centres specialising in maternal and childhood diseases.
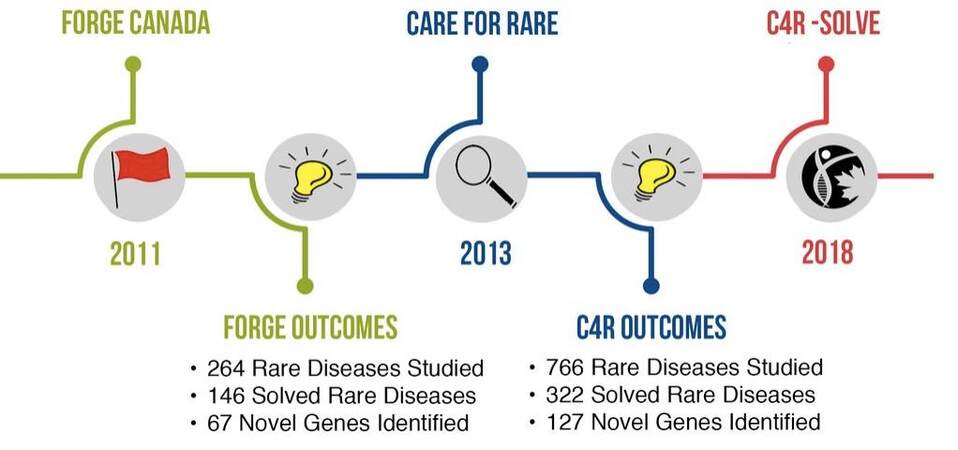
The diagram presented in Figure 4 shows the timeline of research studies conducted by the RDMM as part of the FORGE programme (FORGE: Programme set up to identify the genes causing rare diseases in Canada) which became the international research consortium Care4Rare in 2013.
Figure 4. Timeline of the research studies led by the RDMM (© Copyright 2014. “Rare Diseases: Models & Mechanisms Network” All rights reserved).
(c) Care4Rare (C4R)[14] brings together Canadian clinicians, bioinformaticians, scientists and researchers working on the care of patients with rare diseases in Canada and around the world. Led by the Children’s Hospital of Eastern Ontario (CHEO) and the Ottawa Research Institute in Ottawa, it has 21 academic centres across Canada with 200 physicists and 100 scientists.
(d). C4R-SOLVE[14] uses innovative new sequencing technologies based on omic approaches [Omics technologies: a transdisciplinary approach (chemistry, physics and information technology) based on a systematic analytical approach to the content of living cells at the molecular level], to build an infrastructure with the tools necessary to improve the diagnosis of rare diseases worldwide.
(e). In 2014, the PhenomeCentral system brought together research and diagnosis in a single portal. Developed by the Centre for Computational Medicine at the Hospital for Sick Children in Toronto, this connected system is able to secure data[15]. It provides a global link between patients with identical genotypes and phenotypes, as well as clinicians and scientists working on similar cases. PhenomeCentral is funded by the CIHR IG, the Ontario Genomics Institute, and the Natural Sciences and Engineering Research Council (NSERC). It is a partner of the NIH, RD-Connect (an EU-funded integrated platform) and the International Rare Diseases Research Consortium, funded by the United States and the EU.
(f). Ontario Genomics is a not-for-profit organisation supported by the CIHR IG and the government of Ontario. It brings together companies, researchers, policy makers and funders to generate funding and accelerate the development of genomics[16].
(g). The Canadian Organization for Rare Diseases (CORD) was set up by the Canadian Parliament in 2015[17].
Its main priorities are to:
- Implement a regulatory framework;
- Put in place appropriate funding for equitable patient access to treatment;
- Identify centres of excellence bringing together research, patients and care;
- Set up a programme for the screening of newborn babies.
In 2019, the CIHR announced an investment of €680 million in rare diseases[18]. Primarily dedicated to ensuring patient access to healthcare, this sum will be divided into two equal payments in 2022 and 2023.
6. United States: in 2012, the USA put in place an accelerated registration process for ultra-rare diseases[19].
The Orphan Drug Act of the Food and Drug Administration (FDA) defines a rare disease as a disease or condition that affects less than 200,000 people in the United States.
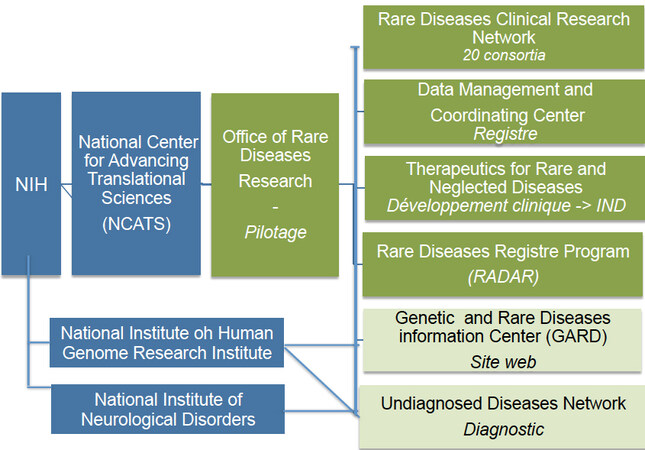
As is often the case in the United States, resources are provided by the national, i.e. Federal government. The National Institutes of Health (NIH) play a key role in the field of rare diseases. As presented in Figure 5, NIH is structured as follows:
(a). The National Center for Advancing Translational Sciences (NCATS) is one of the 27 institutes making up the NIHs. It comprises seven divisions, one of which is dedicated to rare diseases.
The grant received by the NIH in 2020 totalled €722 million, or 0.2% of its budget (€35 billion)[20].
(b). The Office of Rare Diseases Research (ORDR) is the steering organisation of the NCATS Rare Disease Division[21].
(c). Founded in 2003 by the NIH, the Rare Diseases Clinical Research Network (RDCRN) is made up of 20 consortia specialising in clinical research, bringing together scientists, clinicians, patients and associations, i.e. 2600 experts at present[22,23]. Since its founding, the RDCRN has studied over 280 diseases and recruited around 40,000 patients for clinical trials.
(d).The Data Management and Coordinating Centre (DMCC) is based at the Cincinnati Children’s Hospital. It collects, homogenises, manages and shares RDCRN data in order to increase dissemination for research purposes.
(e). The Therapeutics for Rare and Neglected Diseases (TRND) programme is involved from pre-clinical development through to submission of an IND to the FDA. It brings together NIH academic research teams and private partners to accelerate the development of new molecules.
One of the activities of TRND is to seek new applications for compounds that have already passedclinical trial phases[24].
(f). The Genetic and Rare Diseases Information Center (GARD) is a website set up to provide information that can be easily understood by all audiences[25]. GARD is funded by the NCATS and the National Human Genome Research Institute (NHGRI), another department of the NIH. It is connected to Orphanet.
(g). The Rare Diseases Registry Program (RaDaR) is a website dedicated to patients, their families, researchers and the public[26]. It is currently in Phase 1.
(h). The Undiagnosed Diseases Network (UDN) is funded by the National Human Genome Research Institute (NHGRI), the NCATS and the National Institute of Neurological Disorders (NIND)[27]. Its 12 sites bring together clinicians and scientific experts pooling their efforts to accelerate gene identification. Since its launch in 2015, 200 cases have been diagnosed.
The university is the basic entity in research and development, whether clinical (hospitals and medical schools) or fundamental (research laboratories). Most funding is private, with the contribution from the federal government making up around 20% of the total.
Since 2012, the Food and Drug Administration (FDA) has simplified the regulations governing clinical research in the field of rare diseases. Special terms are made available to manufacturers: financial and tax support and accelerated procedures for bringing new products to market.
East Asia
1. Japan: a forerunner in the field of detection, Japan uses innovative technologies for whole genome and exome sequencing (The exome is the part of the genome most directly linked to the phenotype of the organism, to its structural and functional qualities).
In Japan, rare diseases are defined as diseases with a prevalence of less than 50,000 and with no known cause or cure.
Implemented in 2014, the Japanese plan for Rare and Undiagnosed Diseases succeeded in the rare disease research programme set up in 1972 by the Japan Agency for Medical Research and Development (AMED) under the name “Nan-Byo Research”[28,29]. The Ministry of Health is a key player in the new plan, led by AMED. It involves 34 clinical investigation centres and four analysis centres, and funds around 500 doctors and 50 coordinators belonging to the National Coordination Committee (European journal of human genetics 5/7/2017)
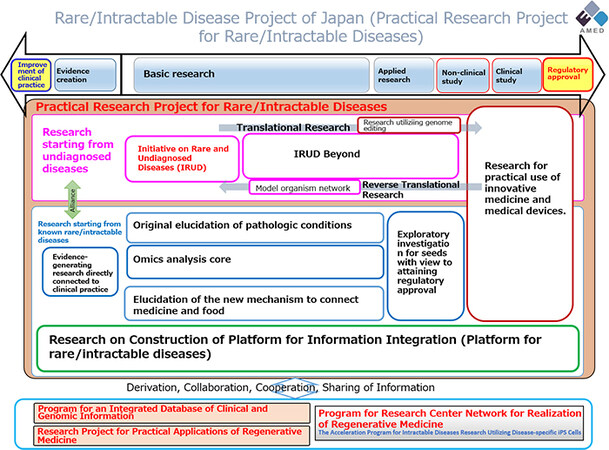
The plan for rare diseases [Figure 6] has two main strands: Decision-making policy for rare diseases and practical applications of the measures adopted[30]. The investment priorities are as follows:
Figure 6. Japan project for rare diseases (Copyright © Japan Agency for Medical Research and Development, All Rights Reserved).
- Pathology and mechanism of rare diseases;
- Repositioning of existing drugs and medical devices for clinical use in patients suffering from rare diseases;
- Improved clinical practices (training) to optimise the effectiveness of healthcare;
- Use of innovative omics technologies;
- Implementation of the Initiative on Rare and Undiagnosed Diseases (IRUD);
- Creation of a data registry compatible with international standards;
- Development of innovative drugs using gene therapy or nucleic acids;
- Production of vectors for the development of new therapies;
- Research into the links between medicine, nutrition and the physiological aspects of rare diseases.
In line with these priorities, AMED has put in place the following approach:
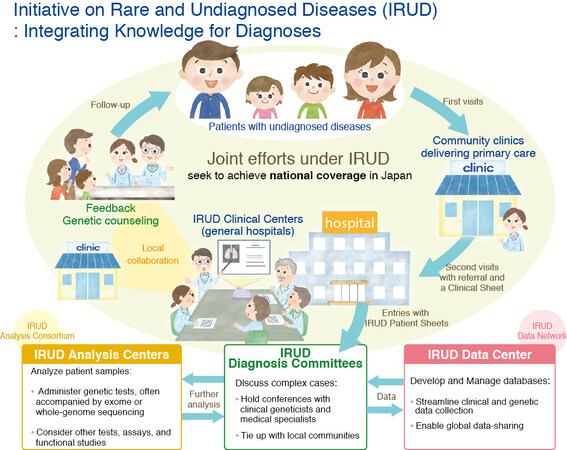
(a). The Initiative on Rare and Undiagnosed Diseases (IRUD) [Figure 7], implemented by AMED in 2015, is part of Japan’s Rare Disease Plan[31]. IRUD is a national consortium that brings together patients, physicians and researchers as part of the process below:
Figure 7. Initiative on Rare and Undiagnosed Diseases (IRUD) (Copyright © Japan Agency for Medical Research and Development, All Rights Reserved).
(b). Launched by AMED in February 2017 and funded by AMED and the Ministry of Health, Labour and Welfare, the National Platform for the Rare Diseases Data Registry of Japan (RADDAR-J) collects and structures data from research projects into rare diseases[32]. The experts involved in the IRUD consortium are connected to this registry, which includes data relating to over 2,000 undiagnosed patients (Source: European Journal of Human Genetics 5/7/2017). The database is designed to be linked to international platforms in compliance with the standards set out by the International Rare Diseases Research Consortium (IRDiRC) consortium as part of the Automatable Discovery and Access-Matrix[33]. In the longer term, AMED is seeking a connection with the international platform of the IRDIRC consortium, scheduled for launch in 2027.
(c). The Office of Pharmaceutical Industry Research (OPIR) was set up in January 1999 by the Japan Pharmaceutical Manufacturers Association (JPMA) (The Japan Pharmaceutical Manufacturers Association (JPMA) represents the research-based pharmaceutical industry in Japan. The JPMA has 74 members including 20 overseas affiliates.). OPIR analyses the internal and external environment of the Japanese pharmaceutical industry (in all fields), identifying the mid- and long-term issues, with a view to submitting recommendations to policy makers[34]. OPIR has developed an economic and social structure that contributes to the rapid development of medical innovations. OPIR is an open laboratory for research, policy recommendations and the development of human resources [Figure 8]. The structure supports its own research projects as well as collaborating with third parties.
CONCLUSION
Outlining an optimal approach
The description of the seven approaches coordinated by governments to support and treat patients with rare diseases [Table 1] shows a number of strengths [Table 2] but also a number of divergences. The approach adopted by each country reflects its history, the commitment of the State, the organisation of the healthcare and biomedical research systems, national legislation and the culture in terms of innovation and public-private partnerships.
A comparison of approaches based on specific key criteria
| Steering | Source of funding | Centres of expertise | Gene identification | National register | |
| Germany 2013 | State + patient organisation (ASCHE) | State + patient organisation (ASCHE) | 36 centres working with 8 research centres + 10 centres for NDD | RD-Connect programme (EU funding 2018-2022): -> register relating to research Biobanks and the creation of an EU platform | Se-Atlas: 8000 genes CORD-MI project €160 million invested between 2016 et 2021 |
| Spain 2009 | State | State | 19 centres of expertise Spain UDP consortium | The Human Genetic Area (AGH) for diagnosed diseases. SpainUDP for undiagnosed diseases | SpainRDR Funding from the State / Regions and collaboration with private partners |
| France 2004 | State | State | 400 reference centres for rare diseases (CRMRs); 27 National Rare Disease Health Networks (FSMRs) | France Genome 2025 programme High-throughput platform | Deployment of the BaMaRa register |
| United Kingdom 2012 | State | State | 76 centres Emphasis on patient care and empowerment | 13 centres, involving 85 hospitals Target 2023: sequencing of 50,000 genes | - |
| Canada 2015 | State: Canadian Organization for Rare Diseases (CORD) | Genome Canada, funded by federal government, the provinces and manufacturers | 20 mother and child research centres | 6 genomic centres Care4Rare, international, transdisciplinary consortium, with 21 sites | PhenomeCentral: national register with international scope |
| USA 1983 | State: NIH | State | Network of 20 consortia, with 350 sites in the United States and 22 worldwide | Undiagnosed Diseases Network (2015), with 12 sites | Data Management and Coordinating Center (DMCC) |
| Japan 1972 | State | State | 34 clinical investigation centres 4 analysis centres | Consortium for the Initiative on Rare and Undiagnosed Diseases (IRUD) | RADDAR platform connected to international platforms |
The strengths of national approaches
| Public/private partnership (PPP) | Key points of national programmes | |
| Germany | 2021, creation of a State-funded programme dedicated to translational medicine and pharmacology (Fraunhofer ITMP): Development of innovative solutions in partnership with industry and training | - PPP: translational research targeting rare diseases: ≥ rationalisation - The Fraunhofer business model has demonstrated its efficiency in other fields |
| Spain | SpainRDR register: collaboration with private partners | - SpainUDP programme, many national and international connections - SpainRDR register: Collaboration with private partners |
| France | 5 Institutes of Excellence (healthcare, teaching, training, translational research). PPP, involves manufacturers from the outset | - Organisation of centres of expertise and healthcare networks for rare diseases. - Strong EU involvement as part of the JPRD - PPP: IHU university-hospital institutes, centres of healthcare excellence and translational research |
| United Kingdom | - Catapult Cell Therapy Centre, private: clinical trials, provision of infrastructure, search for funding from manufacturers. - Stevenage Centre, dedicated to gene and cell therapy manufacturing | - Structure: strong State involvement - Patient healthcare and empowerment - PPP: the CC-CT Catapult Centre involves 64 public and private partners |
| Canada | - Genomic Canada: manages funds from the State, provinces, and industry to invest in PPP projects - Ontario Genomic, supported by the government and State of Ontario brings together PP players to accelerate genomic development | - Private partners involved from the outset - gene identification: international collaboration as part of the Care4Rare consortium. - PhenomeCentral register |
| USA | - Therapeutics for Rare and Neglected Diseases (TRND), the State funds the development of molecules through to the submission of an IND as part of a PPP - Co-funding with industry in Universities | - Structure: Strong State involvement - FDA: Simplified regulations. - State funding -> filing of an IND as part of a PPP - PPP in universities: project research |
| Japan | The Office of Pharmaceutical Industry Research (OPIR) makes recommendations to political decision-makers OPIR collaborates with academic research | - Strong State commitment - Use of innovative technologies for sequencing - Register/Platform compatible with international standards |
Based on a few key criteria necessary for the development of a national system, we have compared the organisation of each country and highlighted a number of specific characteristics.
(a). The government steers and funds all the programmes, sometimes in association with private partners, in Germany with the ASCHE patient organisation and in Canada with the Genome Canada structure.
(b). Significant disparities can be seen in the establishment of centres of expertise:
In Canada and the United States, multidisciplinary research consortia are funded by the State, whereas centres of expertise are housed in hospitals that are mainly private;
In Spain, the SpainUDP translational programme for undiagnosed diseases is coordinated with the 19 centres of expertise;
In France, 400 reference centres look after patients and organise their healthcare, while 23 Health Networks for Rare Diseases bring together the players involved in one or more diseases;
In Japan, the centres of expertise are associated with clinical investigation centres;
In the United Kingdom, the government prioritises investments in patient care and empowerment.
Finally, the number of centres is not proportional to population density.
(c). In terms of genomics, Canada and Japan are pioneers, Japan as early as 1972 with the Nan-Byo programme, and Canada from 2000 with Genome Canada.
(d). International collaboration is a key benefit that is very much a part of the system in North America, Spain and Japan.
(e). The state of development of national registry programmes varies considerably. Canada, Spain, the United States, France and Japan have operational tools Canada, Spain and the United States are connected to the Undiagnosed Diseases Network International (UNDI) (UNDI partner countries: Australia, Austria, Belgium, Bulgaria, Canada, Hungary, India, Italy, Korea, Spain, Sri Lanka, Sweden, Thailand, Turkey, USA.) consortium, sharing their data for research purposes.
(f). Depending on the country, industrial partners may be involved at different stages of the plan:
- In Germany and the United Kingdom, the public-private centres specialised in a particular field, such as the Fraunhofer ITMP and the CGT Catapult, seek to promote industrial partnerships;
- In Canada, private partners are associated with the State from the outset;
- In Spain and France, private partnerships are mainly based on public university hospital campuses. In France, the launch in 2010 of University Hospital Institutes (IHU), jointly funded by government and industry, encourages public/private partnerships.
- In the United States, the TRND co-funds the development of drugs with manufacturers until an IND is filed. Moreover, 80% of university research (all fields) is funded by private partnerships;
- In Japan, OPIR makes policy recommendations, supports research projects and sets up joint projects with third parties.
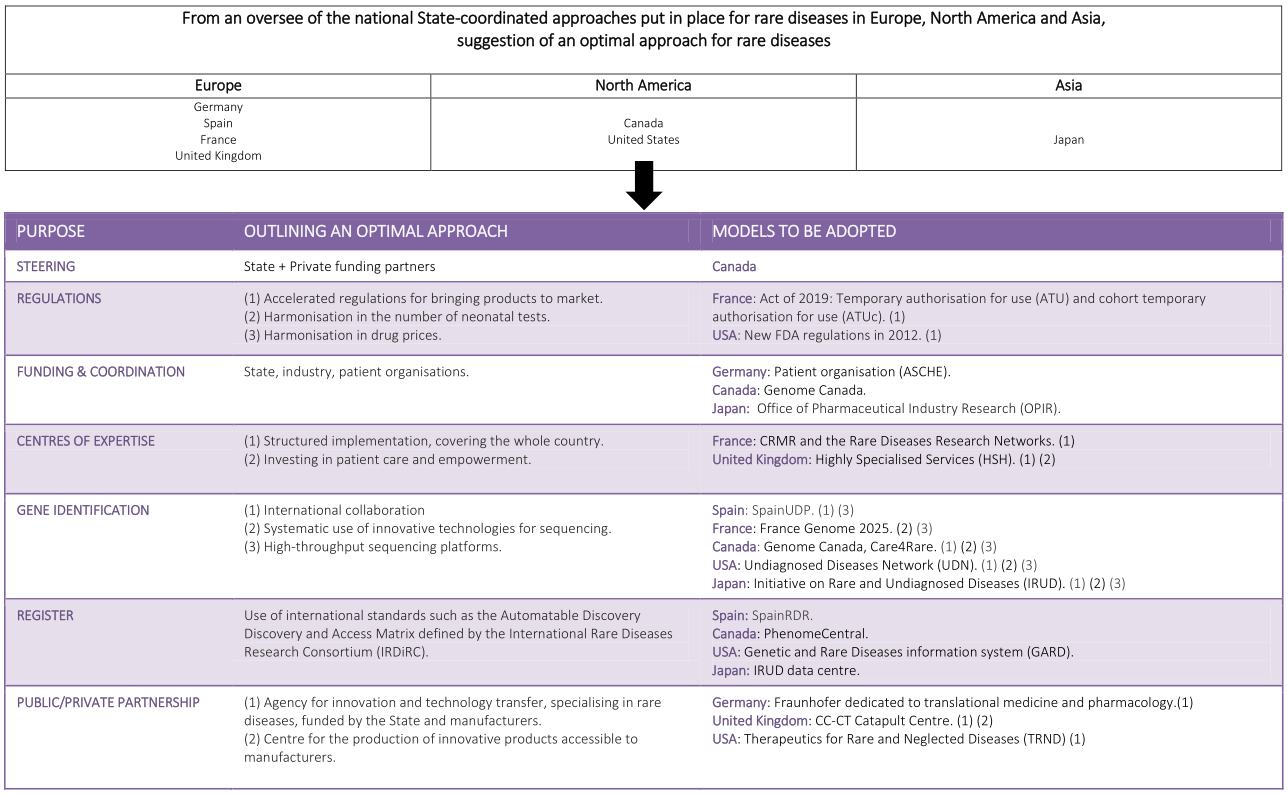
In conclusion, inspired by the successes of the different organisations of the countries previously presented, an optimal approach could be suggested [Table 3], avoiding pitfalls. In this approach:
A possible outline for an optimal approach
| Purpose | Outlining an optimal approach | Models to be adopted |
| Steering | State + Private funding partners | Canada |
| Regulations | (1) Accelerated regulations for bringing products to market (2) Harmonisation in the number of neonatal tests (3) Harmonisation in drug prices | France: Act of 2019: Temporary authorisation for use (ATU) and cohort temporary authorisation for use (ATUc) (1) USA: New FDA regulations in 2012 (1) |
| Funding & coordination | State, industry, patient organisations | Germany: patient organisation (ASCHE) Canada: genome Canada Japan: Office of Pharmaceutical Industry Research (OPIR) |
| Centres of expertise | (1) Structured implementation, covering the whole country (2) Investing in patient care and empowerment | France: CRMR and the Rare Diseases Research Networks (1) United Kingdom: Highly Specialised Services (HSH) (1) (2) |
| Gene identification | (1) International collaboration (2) Systematic use of innovative technologies for sequencing (3) High-throughput sequencing platforms | Spain: SpainUDP (1) (3) France: France Genome 2025 (2) (3) Canada: Genome Canada, Care4Rare (1) (2) (3) USA: Undiagnosed Diseases Network (UDN) (1) (2) (3) Japan: Initiative on Rare and Undiagnosed Diseases (IRUD). (1) (2) (3) |
| Register | Use of international standards such as the Automatable Discovery and Access Matrix defined by the International Rare Diseases Research Consortium (IRDiRC) | Spain: SpainRDR Canada: PhenomeCentral. USA: Genetic and Rare Diseases information system (GARD) Japan: IRUD data centre |
| Public/private partnership | (1) Agency for innovation and technology transfer, specialising in rare diseases, funded by the State and manufacturers (2) Centre for the production of innovative products accessible to manufacturers | Germany: Fraunhofer dedicated to translational medicine and pharmacology (1) United Kingdom: CC-CT Catapult Centre (1) (2) USA: Therapeutics for Rare and Neglected Diseases (TRND) (1) |
√ The government and private partners (on a pro rata basis according to their participation) would steer and fund the system, in the same way as with the Genome Canada model.
√ The organisation of the centres of expertise would be based on the French model with increased financial support for patient empowerment (UK).
√ In terms of genomics, the emphasis would be placed on international collaboration and information sharing, with involvement in international consortia such as Care4Rare.
√ The ongoing development of national registries would be based on international standards, for connection to the European and international platforms currently under development.
√ The involvement of manufacturers from the outset in order to favour partnerships (Canada). Furthermore, the results of the CGT Catapult in the United Kingdom underline the relevance of a specialised system dedicated to rare diseases. Finally, the business model of the French IHU has produced real economic benefits and deserves further development.
DISCUSSION
This work examined the different national plans and strategies dedicated to rare diseases in Germany, Spain, the United Kingdom, France, Japan, Canada and the United States. We were especially interested in the commitment of these countries, their healthcare and biomedical research system organisation, their national legislation and their culture in terms of innovation and public-private partnerships. By concentrating the available information on the national plans and strategies, we were able to identify their strengths and divergences, and then define a possible optimal approach [Table 3].
It is important to stress that the 7 countries that are included in our work are not the only countries in Europe, East Asia and North America to have established a national plan or strategy dedicated to rare diseases.
Indeed, regarding the European Union, there are currently a total of 25 Members States that already have established a national plan or strategy [Figure 1], while only 3 of them have been included in our work. Therefore, the optimal approach we have defined could certainly be refined considering the potential successful organisations that are present in the 22 Member States not included. Also, the impact on European countries of the European Union’s policy on rare diseases has not been discussed, but its analysis could provide a complement for our optimal approach.
With the same principle, the national plans and strategies for rare diseases established in East Asian and North American countries, which are not included in our work, could also refine the proposed optimal approach.
Nevertheless, we believe that our work offers an important overview of successful national strategies and plans dedicated to rare diseases from which countries could potentially benefit.
DECLARATIONS
Acknowledgements
OrphanDev (https://www.orphan-dev.org/) is a French national network funded by F-CRIN aiming to bring solutions to the patients suffering from rare diseases. The authors would like to thank F-CRIN for their support.
Authors’ contributions
Conducted the review: Trentesaux V
Discussion and writing of the manuscript: Cortial L, Nguyen C, Julkowska D, Cocqueel-Tiran F, Montserrat Moliner A, Blin O, Trentesaux V
Availability of data and materials
Not applicable.
Financial support and sponsorship
This work was supported by the French Clinical Research Infrastructure Network (F-CRIN).
Conflict of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2022.
REFERENCES
1. NAMSE. National action league for people with rare diseases. Available from: https://www.namse.de/english [Last accessed on 23 May 2022].
2. Haase J, Wagner TOF, Storf H. [se-atlas - the health service information platform for people with rare diseases : Supporting research on medical care institutions and support groups]. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz 2017;60:503-9.
3. Medical Informatics Initiative. About the initiative. Available from: https://www.medizininformatik-initiative.de/index.php/en/about-initiative [Last accessed on 23 May 2022].
4. Institute - Fraunhofer ITMP. Fraunhofer institute for translational medicine and pharmacology ITMP. Available from: https://www.itmp.fraunhofer.de/en/institute.html [Last accessed on 23 May 2022].
5. Fraunhofer-Annual-Report-2019. Available from: https://www.fraunhofer.de/content/dam/zv/en/Publications/Annual-Report/2019/Fraunhofer-Annual-Report-2019.pdf [Last accessed on 23 May 2022].
6. Pages - Our ISCIII Centres. Available from: https://eng.isciii.es/eng.isciii.es/QuienesSomos/CentrosPropios/Paginas/default.html [Last accessed on 23 May 2022].
7. NHS commissioning. Highly specialised services. Available from: https://www.england.nhs.uk/commissioning/spec-services/highly-spec-services/ [Last accessed on 23 May 2022].
8. Our Centres. The catapult network. Available from: https://catapult.org.uk/about-us/our-centres/ [Last accessed on 23 May 2022].
9. Cell and Gene Therapy Catapult - Annual Reviews. Available from: https://ct.catapult.org.uk/our-impact/annual-reviews [Last accessed on 23 May 2022].
10. Construction completed on CGT Catapult expansion. Available from: https://ct.catapult.org.uk/news-media/manufacturing-news/construction-completed-expansion-phase-doubling-capacity [Last accessed on 23 May 2022].
11. Genome Canada. Genome Canada’s role. Available from: https://www.genomecanada.ca/en/why-genomics/genome-canadas-role [Last accessed on 23 May 2022].
12. Indigenous Health. The silent genomes project. Available from: https://www.indigenoushealthnh.ca/news/silent-genomes-project-indigenous-DNA [Last accessed on 23 May 2022].
13. Rare diseases: models & mechanisms network. Available from: http://www.rare-diseases-catalyst-network.ca/about-the-network.html [Last accessed on 17 Jan 2022].
14. CARE for RARE. Available from: http://care4rare.ca [Last accessed on 17 Jan 2022].
15. Buske OJ, Girdea M, Dumitriu S, et al. PhenomeCentral: a portal for phenotypic and genotypic matchmaking of patients with rare genetic diseases. Hum Mutat 2015;36:931-40.
16. Ontario genomics. What we do. Available from: https://www.ontariogenomics.ca/about-us/what-we-do/ [Last accessed on 23 May 2022].
17. CORD. Now is the time: a strategy for rare diseases is a strategy for all Canadians. Available from: http://www.raredisorders.ca/content/uploads/CORD_Canada_RD_Strategy_22May15.pdf [Last accessed on 23 May 2022].
18. Government of Canada. National strategy for drugs for rare diseases online engagement. Available from: https://www.canada.ca/en/health-canada/programs/consultation-national-strategy-high-cost-drugs-rare-diseases-online-engagement.html [Last accessed on 23 May 2022].
19. FDA. Developing Products for Rare Diseases & Conditions. Available from: https://www.fda.gov/industry/developing-products-rare-diseases-conditions [Last accessed on 17 Jan 2022].
20. Congressional Research Service. National institutes of health (NIH) funding: FY1996-FY2023. Available from: https://sgp.fas.org/crs/misc/R43341.pdf [Last accessed on 23 May 2022].
21. GARD. Office of Rare Diseases Research (ORDR) Brochure - Office of Rare Diseases Research. Available from: https://rarediseases.info.nih.gov/asp/resources/ord_brochure.html [Last accessed on 23 May 2022].
22. Rare Diseases Clinical Research Network. Available from: https://www.rarediseasesnetwork.org/ [Last accessed on 23 May 2022].
23. National Center for Advancing Translational Sciences. Rare diseases clinical research network (RDCRN). Available from: https://ncats.nih.gov/rdcrn [Last accessed on 23 May 2022].
24. National Center for Advancing Translational Sciences. Therapeutics for rare and neglected diseases (TRND). Available from: https://ncats.nih.gov/trnd [Last accessed on 18 Jan 2022].
25. Genetic and Rare Diseases Information Center (GARD) - an NCATS Program. Available from: https://rarediseases.info.nih.gov/ [Last accessed on 23 May 2022].
26. National center for advancing translational sciences. Rare diseases registry program (RaDaR). Available from: https://ncats.nih.gov/radar [Last accessed on 23 May 2022].
27. UDN. Undiagnosed diseases network. Available from: https://undiagnosed.hms.harvard.edu/ [Last accessed on 23 May 2022].
28. Ito T. Patient groups’ advocacy activity and nanbyo act (The Iatric Services to Patients Suffering from Intractable Disease): from the view point of patient groups. Journal of Health Care and Society 2018;28:27-36.
29. Richter T, Nestler-Parr S, Babela R, et al. International Society for Pharmacoeconomics and Outcomes Research Rare Disease Special Interest Group. Rare disease terminology and definitions-a systematic global review: report of the ISPOR rare disease special interest group. Value Health 2015;18:906-14.
30. Japan Agency for Medical Research and Development. Practical research project for rare / intractable diseases. Available from: https://www.amed.go.jp/en/program/list/11/02/003.html [Last accessed on 23 May 2022].
31. Adachi T, Kawamura K, Furusawa Y, et al. Japan’s initiative on rare and undiagnosed diseases (IRUD): towards an end to the diagnostic odyssey. Eur J Hum Genet 2017;25:1025-8.
32. RADDAR-J. RADDAR-J - Rare Disease Data Registry of Japan. Available from: https://www.raddarj.org/en/ [Last accessed on 18 Jan 2022].
33. IRDiRC. Automatable discovery and access. Available from: https://irdirc.org/activities/task-forces/automatable-discovery-and-access/ [Last accessed on 18 Jan 2022].
34. About OPIR. Office of pharmaceutical industry research. Available from: https://www.jpma.or.jp/opir/en/about/index.html [Last accessed on 18 Jan 2022].
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Cortial L, Nguyen C, Julkowska D, Cocqueel-Tiran F, Moliner AM, Blin O, Trentesaux V. Managing rare diseases: examples of national approaches in Europe, North America and East Asia. Rare Dis Orphan Drugs J 2022;1:10. http://dx.doi.org/10.20517/rdodj.2022.02
AMA Style
Cortial L, Nguyen C, Julkowska D, Cocqueel-Tiran F, Moliner AM, Blin O, Trentesaux V. Managing rare diseases: examples of national approaches in Europe, North America and East Asia. Rare Disease and Orphan Drugs Journal. 2022; 1(2): 10. http://dx.doi.org/10.20517/rdodj.2022.02
Chicago/Turabian Style
Cortial, Lucas, Catherine Nguyen, Daria Julkowska, Florence Cocqueel-Tiran, Antoni Montserrat Moliner, Olivier Blin, Valérie Trentesaux. 2022. "Managing rare diseases: examples of national approaches in Europe, North America and East Asia" Rare Disease and Orphan Drugs Journal. 1, no.2: 10. http://dx.doi.org/10.20517/rdodj.2022.02
ACS Style
Cortial, L.; Nguyen C.; Julkowska D.; Cocqueel-Tiran F.; Moliner AM.; Blin O.; Trentesaux V. Managing rare diseases: examples of national approaches in Europe, North America and East Asia. Rare. Dis. Orphan. Drugs. J. 2022, 1, 10. http://dx.doi.org/10.20517/rdodj.2022.02
About This Article
Copyright
Data & Comments
Data
 Cite This Article 27 clicks
Cite This Article 27 clicks













Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.